Preterm birth is associated with mortality and long-term disabilities. Despite the advances in neonatal medicine in the last 20 years, brain injury remains a burden for those infants who survive. Non-invasive methods based on near-infrared spectroscopy (NIRS) brain monitoring could potentially be used for optimising neonatal care and improving neurological outcome for preterm infants, including determination of optimal blood pressure, avoiding ischemia, or the risk of brain haemorrhage. ICM+ provides tools for exploration and development of those ideas.
Brain protection of premature newborns is an emerging subject in neonatal intensive care. Survival is much better than 10 years ago, but a level of secondary brain damage should be minimised.
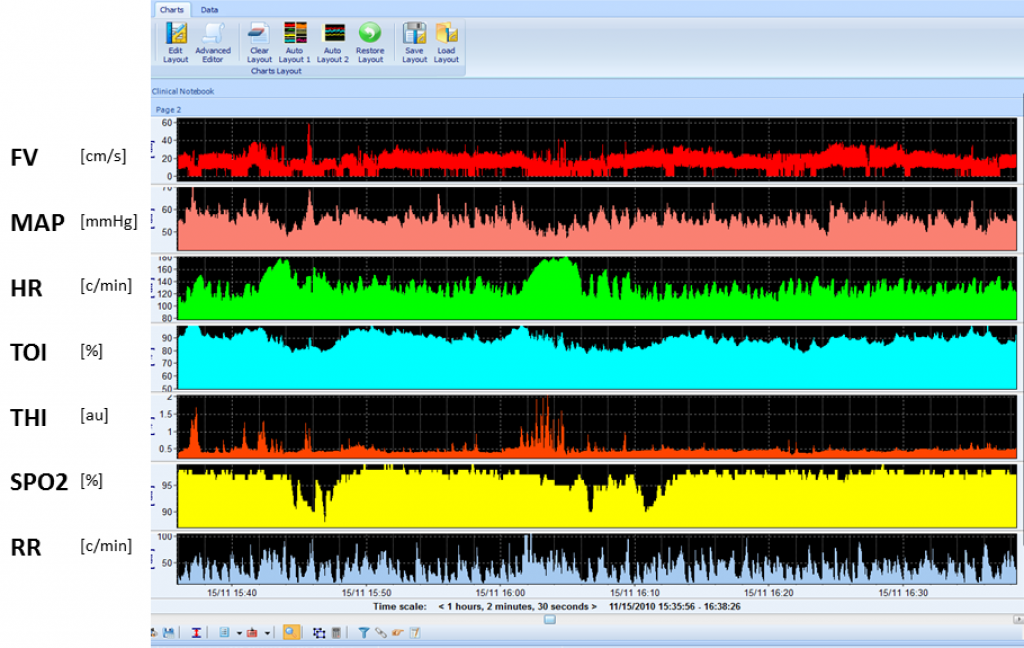
Monitoring of brain in premature neonates mainly utilises non-invasive modalities like TCD and NIRS. SPO2; non-invasive or invasive arterial pressure are commonly included. View image
Optimal ABP determination
Changes in brain oxygenation in premature babies essentially reflect changes in CBF to the brain. By comparing changes in the baby’s arterial blood pressure (ABP) with changes in cerebral blood flow (CBF), it is possible to get information on how well the cerebral circulation is being regulated – a process known as cerebral autoregulation. In healthy babies, CBF remains relatively constant over a wide arterial blood pressure (ABP) range, however, in sick infants, this control is lost and any fluctuations in ABP are probably transmitted to the brain. This leaves the brain vulnerable to low (ischaemia) and high CBF (intraparenchymal haemorrhage).
In recent research using ICM+, an ABP based on an index of cerebrovascular reactivity (TOHRx) has been determined. TOHRx is known as the tissue oxygenation-heart rate reactivity index, and describes cerebral vascular reactivity using the correlation coefficient between slow waves of tissue oxygenation index (TOI, measured with NIRS) and the heart rate (HR). Impaired vascular reactivity (positive values of TOHRx) has been associated with higher CRIB II scores (clinical risk index for baby). Optimal ABP represents the value where cerebrovascular reactivity is the strongest. Preterm infants who died or had worse germinal matrix-intraventricular haemorrhages have presented with a higher mean absolute deviation from optimal ABP than patients who survived.
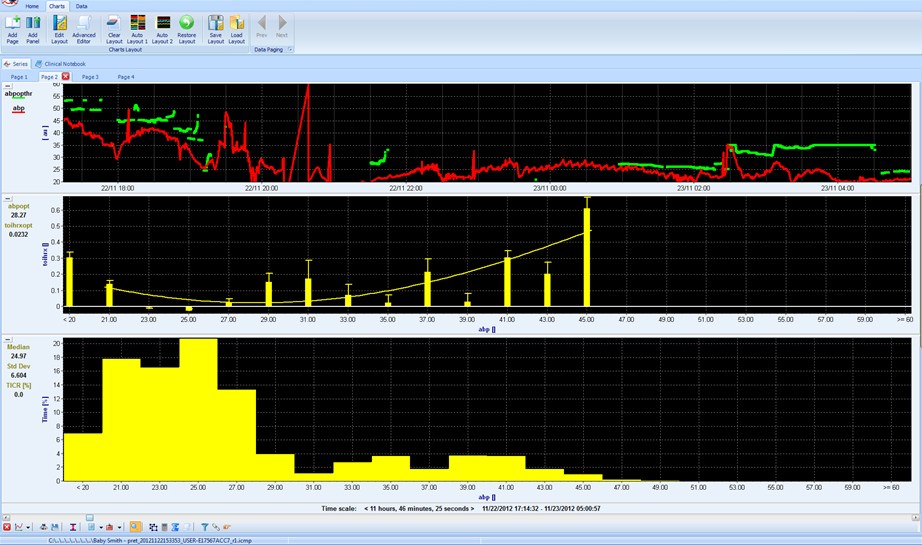
Optimal ABP: ABP at which TOIHRx reaches minimum. This point can be found in about 2/3 of the patients. View image
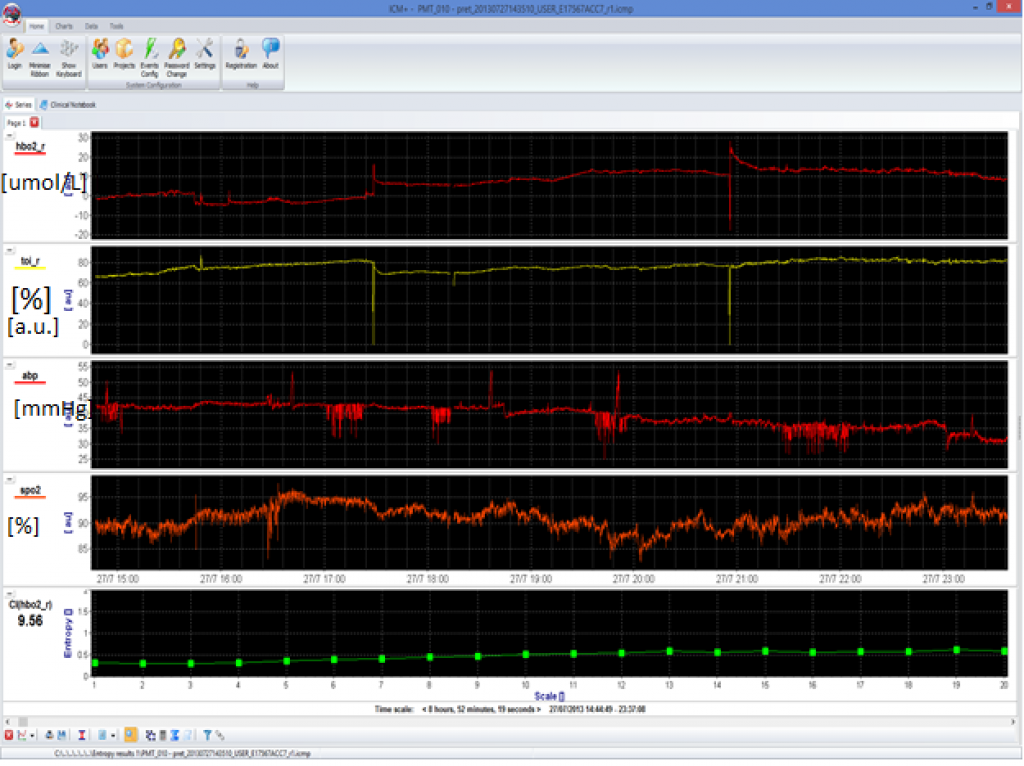
Complexity of oxegenated hemoglobin can be studied. It has been proven that low complexity is associated with mortality of preterm newborns. View image

